Laboratory of Oncology
Projects in the Laboratory of Oncology
In order to develop novel and effective therapeutic strategies to cure cancer, we aim to:
- Understand the molecular mechanisms underlying EndMT which is involved in cancer
progression. - Elucidate the molecular mechanisms underlying tumor angio- and lympangiognesis.
- Develop fully retargeted herpes simplex virus (HSV) vectors for oncolytic virotherapy,and
- Establish highly functional monoclonal antibodies through novel screening methods
for targeted cancer therapy.
Understanding the molecular mechanisms underlying EndMT which is involved in
cancer progression
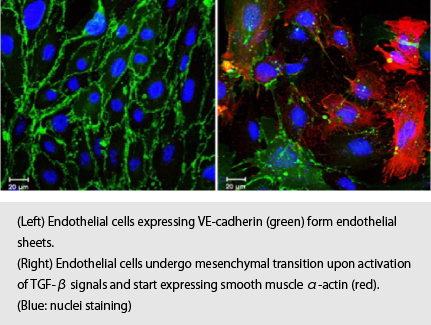
Epithelial-mesenchymal transition (EMT) plays important roles in various physiological and pathological processes, and is regulated by signaling pathways mediated by cytokines including transforming growth factor (TGF)-β. Endothelial cells also undergo differentiation into mesenchymal cells during not only various physiological processes including heart valve formation but also pathological processes including cancer progression, heart failure and diabetes. However, the molecular mechanisms that regulate such endothelial-mesenchymal transition (EndMT) remain to be elucidated. We aim to study the molecular mechanisms underlying EndMT in order to identify novel targets and attempt to develop therapeutic strategies for EndMT-related diseases.
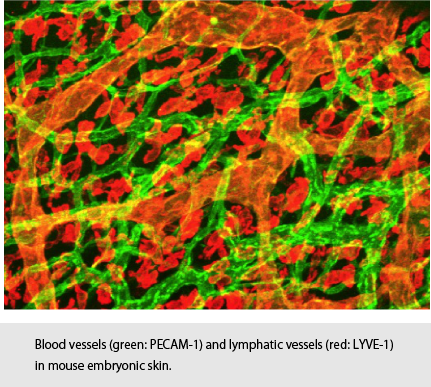
Elucidation of the molecular mechanisms underlying tumor angio- and lympangiognesis
Tumor angiogenesis and lymphangiogenesis are key features of tumor progression and metastasis. While multiple signaling pathways have been implicated in the formation of blood and lymphatic vessels, the molecular mechanisms underlying these processes have not yet fully elucidated. Recent findings revealed that members of the transforming growth factor-β (TGF-β) family play pivotal roles on in angiogenesis and lymphangiogenesis, and that abnormalities in TGF-β family signaling lead to development of certain vascular disorders, including hereditary hemorrhagic telangiectasia (HHT), pulmonary arterial hypertension, Marfan syndrome and Loeys-Dietz syndrome. We attempt to elucidate the molecular mechanisms how TGF-β family signals regulate antiogenesis and lymphaniogenesis in tumor microenvironment.
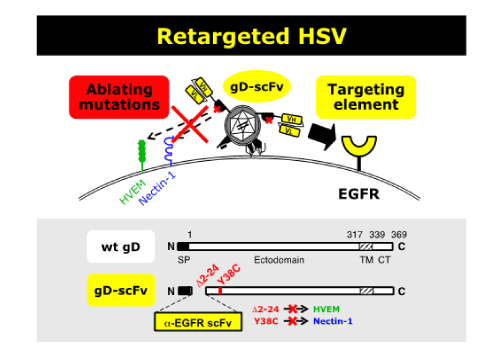
Development of fully retargeted herpes simplex virus (HSV) vectors for oncolytic virotherapy
Herpes simplex virus (HSV) vectors are promising agents for oncolytic virotherapy. Uchida established a fully retargeted HSV platform that mediates virus entry exclusively via tumor-associated antigens in the lab of Prof. Joseph Glorioso at the University of Pittsburgh. Entry of HSV is initiated by the binding of glycoprotein D (gD) to one of its receptors, herpesvirus entry mediator (HVEM) or nectin-1. This interaction results in a conformational change in gD, triggering sequential activation of gH and gB to execute fusion between the viral envelope and cell membranes. We inserted a number of different single-chain antibodies (scFv) into the retargeted HSV platform that encodes a gD ablated for binding to natural receptors and a gB containing entry-enhancing mutations we previously identified. As a result, we observed specific virus entry into cells expressing the cognate target antigen for each of the retargeted constructs. Our results indicate the adaptability of our system to different targeting ligands, leading to a new generation of broadly applicable and effective oncolytic HSV vectors.
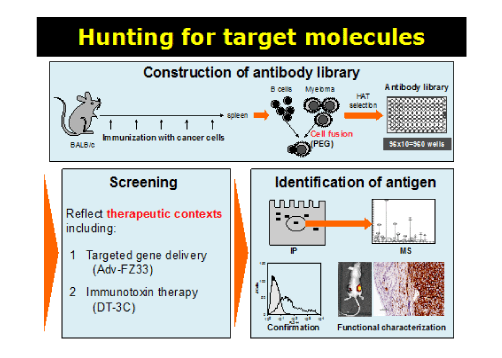
Establishment of highly functional monoclonal antibodies through novel screening methods for
targeted cancer therapy
Monoclonal antibodies (mAbs) have become an established therapeutic modality in clinical oncology. In order to identify cell-surface molecules that may be useful for targeting various types of cancers, our group established a unique screening approach that employs an adenoviral vector harboring fiber proteins engineered to bind antibodies, Adv-FZ33. This approach led to the successful identification of an array of potential target molecules for cancer treatment. Immunotoxins (antibody-drug conjugates; ADC) are a promising class of cancer therapeutics composed of a cytotoxic agent linked covalently to a cancer-targeted antibody. To systematically hunt for cell-surface molecules that may be efficiently targeted by immunotoxins, our group created another method for screening highly functional cancer-targeted mAbs and cognate antigens. The receptor-binding domain of the Diphtheria toxin (DT) was replaced with the antibody-binding domain (3C) derived from the Streptococcal protein G. The resultant mutated toxin protein (DT-3C) was used for selection of mAbs for specific cell killing activity as components of immunotoxins. Our novel screening system is advantageous in that the selected antibodies bind to intact cancer cells with high affinity and get internalized efficiently, which has been critically required for therapeutic applications but elusive thus far.